Numerical Simulation of Flow Electrolysers: Effect of Various Geometric Parameters
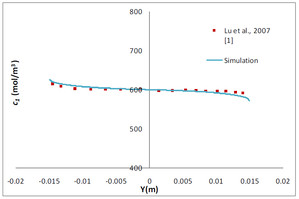
Flow electrolysers find several applications in industry. They are used for production of metals and synthesis of chemicals, gases. Cleaning and preservation of old artifacts, electrolytic refining of metals, electrolytic winning of metals, alkaline water electrolysis, anodization, electrometallurgy, electroplating, electrolytic etching of metal surfaces are other industrial applications of flow electrolysers. Due to their several industrial applications it is important to develop a fundamental understanding of the working of flow electrolysers. This understanding will eventually lead to optimum and efficient designs. The objective of this work is to develop an understanding of coupled phenomena of electrolysis and fluid flow in flow electrolysers having stationary electrodes and to see the effect of various geometrical changes. Two-dimensional numerical simulations have been carried out using COMSOL Multiphysics 3.5a. The present work is aimed at studying 1) effect of inlet channel length, 2) effect of offset between anode and cathode and 3) effect of size of anode on the performance of a flow electrolyser. A simple geometry i.e. rectangular computational domain of length 50 cm and width 10 cm is chosen. The electrolyte considered is NaCl in water. To see the effect of length of inlet channel, simulations have been carried out for different lengths of inlet channel ranging from 20% to 100% of main channel length. The results of simulations for current density equal to 10,000 A/m2 are shown in Fig. 1. The results indicate there is no benefit beyond a certain length of the inlet channel. Figure1 also shows that it is important to consider both Nernst Planck and Navier Stokes equations while simulating flow electrolysers. To see the effect of offset between the anode and cathode a geometry in which anode length was 20 cm and cathode length was 50 cm considered. Figure2 shows the effect of the offset between the anode and cathode on the potential difference across the electrodes. As expected, as the offset increases the potential difference across the electrode increases for a given current density. It is therefore better to design an electolyser in which anode and cathode are of different length such that the offset between the anode and cathode is kept minimum. Figure 3 shows the effect of anode length on the potential difference across the electrodes for a constant total current. The offset between the anode and cathode is zero for these simulations. Fig. 3 shows that as the length of anode reduces, the potential difference between the electrode increases. This is because on reducing the electrode length, the area available for current to traverse between the anode and cathode reduces leading to increased effective resistance. The simulations reported in this work provide useful insight into how the performance of a flow electrolyser is affected when certain geometric parameters namely inlet channel length, offset between anode and cathode and the length of anode are changed. These multiphysics simulations also highlight that it is important to consider both Nernst Planck and Navier Stokes equations while simulating electroneutral bulk of a flow electrolyser.
Download
- shukla_presentation.pdf - 1.06MB
- shukla_paper.pdf - 0.54MB
- shukla_abstract.pdf - 0.1MB
