
In early 2020, a viral video showing an eye-catching lip gloss trick appeared on web browsers and phone screens around the world. The trick features lip gloss mysteriously flying into the air after being pulled from its tube-shaped container, making it look like the lip gloss defies gravity. After a little internet digging, we were able to find a lip gloss that performs the trick. To help further explain how this phenomenon occurs, we can turn to multiphysics simulation…
Explaining a Viral Trend with Simulation
Watch the “magic” lip gloss in action below, and then get an overview of what we think is causing this phenomenon.
Continue reading to learn more about this captivating cosmetic…
Finding a Gravity-Defying Lip Gloss
Acquiring the gravity-defying lip gloss was relatively easy. After testing out a handful of different glosses in a variety of shades and shines, we were able to get our hands on one that successfully and (consistently) demonstrates this trick. In order to prove that the gloss wasn’t just defective, we got four more of the same kind (a little excessive, but it’s in the name of science!) All five lip glosses demonstrate the exact same upward dripping effect.
Tin Oxide: A Likely Culprit
Why do only certain lip glosses perform the gravity trick? A major reason has to do with the ingredients.
The average cosmetic contains anywhere from 15 to 50 ingredients.
After finding two different glosses that both consistently performed the floating phenomenon, we noticed they share a common ingredient: tin oxide. The Cosmetic Ingredient Review (CIR) defines tin oxide as an abrasive, bulking, and opacifying agent in cosmetics. The CIR also reports that tin oxide is safe to use in concentrations up to 1.3% in leave-on products. In its commercial form, it’s manufactured from tin metal by thermal oxidation.

Tin oxide in its mineral form cassiterite, or SnO2. Image in the public domain, via Wikimedia Commons.
Tin oxide is used in lip gloss because it adds a pearlescent shine, decreases density, and helps control viscosity.
It’s important to note that tin oxide concentration isn’t the only reason why the glosses display the trick; it also has to do with their container sizes, applicator shapes, and other ingredients.
Investigating the Lip Gloss Phenomenon with Simulation
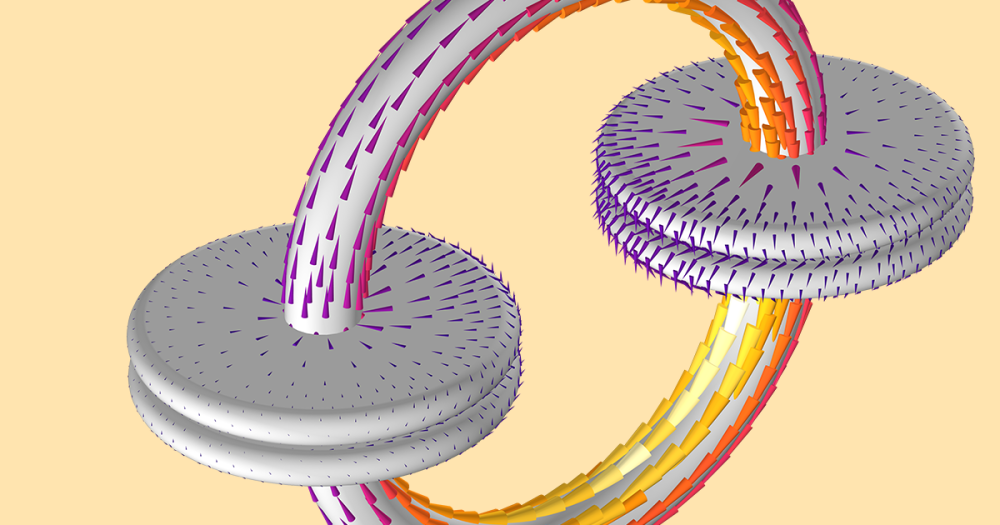
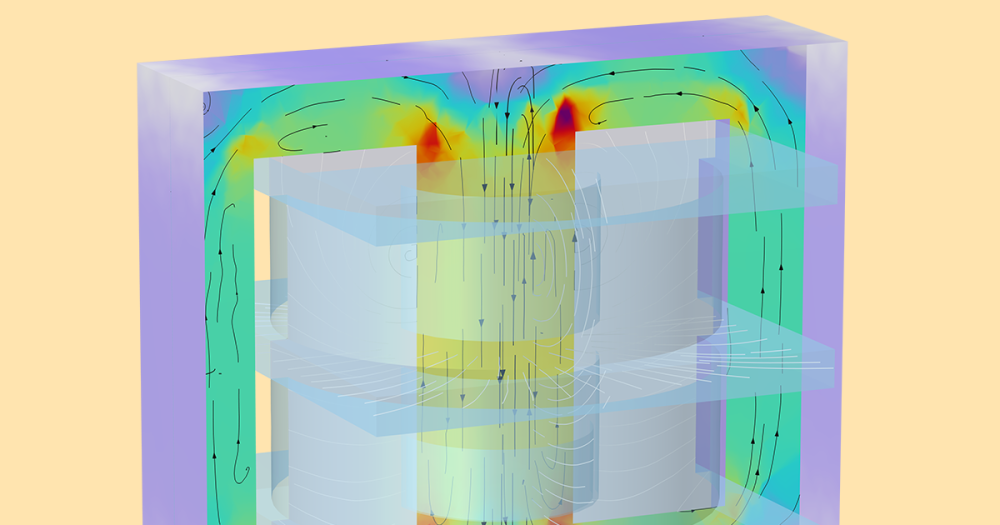
Our theory of why the lip gloss floats? The tin oxide particles inside the gloss’s container rub against each other due to velocity gradients and viscous effects, which creates friction. As the lip gloss’s applicator moves inside and is then separated from its container, the present friction causes a potential difference between the lip gloss in the container and the gloss on the applicator. Between the applicator and the container, an electric field is created. This field causes a driving force for the lip gloss on the applicator to fly toward the container, along the electric field lines.
The polarizations of the tin oxide (or SnO2 nanoparticles) is very high, which enhances the gloss’ dielectric behavior. (Dielectric materials are nonconductive, but they efficiently support electrostatic fields.) The gloss’ dielectric properties give a large-enough charge separation to continue the gravity-defying effect for several minutes.
Electrostatics in Dielectric Material
Considering that the lip gloss is dielectric in nature, let’s briefly go over electrostatics in dielectric materials at the microscopic level…
Electrostatics in idealized dielectric materials have bound charges, which can be replaced by an external electric field. This replacement results in induced electric dipoles within the dielectric material. These dipoles are pairs of positive and negative charges that in some way align with the electric field. The final result? An electric field forms inside the dielectric material (separate from that of free space).
A recent study found that friction (like that caused by the tin oxide particles mixing together in the tube) can cause static electricity by bending the tiny protrusions on the surface of materials.
Solving One Last Lip Gloss Mystery…
You might be wondering what happens to gloss that doesn’t make it back into its container. Unfortunately, it doesn’t magically disappear into the air. As the saying goes: What goes up must come down…

The gloss that didn’t make it back into the container fell onto the ground and the counter.
Further Reading
See more examples of real-world phenomena explained using simulation in these blog posts:




Comments (1)
OLESYA LUPOVICH KULESHOVA
October 3, 2022Hello, I would like to study this so interesting process. My goal: try to write a simple analytical model for this motion, but you have a numerical model. Can you send this Comsol file to me, please?